Indice del volumen Volume index
Comité Editorial Editorial Board
Comité Científico Scientific Committee
HISTOLOGICAL STUDIES OF THE
EFFECTS OF MONOSODIUM GLUTAMATE
ON THE INFERIOR COLLICULUS
OF ADULT WISTAR RATS.
A.O. Eweka, F.A.E. Om'Iniabohs
Department of Anatomy. School of Basic Medical Sciences.
College of Medical Sciences, University of Benin.
Benin City. Edo State. Nigeria.
andreweweka @ yahoo.com
Rev Electron Biomed / Electron J Biomed 2008;3:24-30.
Comment of the reviewer Maxim V. Trushin, PhD. Laboratory of Pathogenesis. Kazan Institute of Biochemistry and Biophysics. Russian Academy of Sciences. Kazan Russia
Comment of the reviewer Rodrigo Valdés Annunziata MD. Centro de Diagnóstico Histopatológico y Citopatológico Histonor Ltda. Antofagasta, Chile.
ABSTRACT:
Histological effects of Monosodium glutamate (MSG) commonly used as food additive on the inferior colliculus (IC) of adult Wistar rats were carefully studied. The rats of both sexes (n=24), average weight of 185g were randomly assigned into two treatments (n=16) and control (n=8) groups. The rats in the treatment groups received 3g and 6g of MSG thoroughly mixed with their feeds for fourteen days, while the control rats received equal amounts of feeds without MSG added. The rats were fed with growers' mash purchased from Edo Feeds and Flour Mill Ltd, Ewu, Edo State and were given water liberally. The rats were sacrificed on day fifteen of the experiment. The inferior colliculus was carefully dissected out and quickly fixed in 10% formal saline for routine histological study after H&E method.
The histological findings after H&E methods indicated that the treated sections of the inferior colliculus showed some cellular degenerative changes, cellular hypertrophy, and autophagic vacuoles with some intercellular vacuolations appearing in the stroma, and some degree of neuronal hypertrophy when compared to the control sections.
These findings indicate that MSG consumption may have a deleterious effect on the neurons of the inferior colliculus (IC). MSG may probably have adverse effects on the auditory sensibilities by its deleterious effects on the nerve cells of the IC of adult Wistar rats. It is recommended that further studies aimed at corroborating these observations be carried out.
Key words: Monosodium glutamate. Histological effect. Inferior colliculus. Intercellular vacuolations. Wistar rats.
RESUMEN: ESTUDIO HISTOLÓGICO DE LOS EFECTOS DEL GLUTAMATO MONOSÓDICO EN EL COLLICULUS INFERIOR DE RATAS WISTAR ADULTAS
Han sido estudiados los efectos histológicos del glutamato monosódico (MSG), comúnmente utilizado como aditivo alimentario, sobre el colliculus inferior (IC) de ratas Wistar adultas. Las ratas de ambos sexos (n = 24), promedio bruto 185g de peso fueron asignadas aleatoriamente a dos pautas de tratamiento (n = 16) y control (n = 8). Las ratas en los grupos de tratamiento recibieron 3g y 6 g de MSG, bien mezclado con la alimentación durante catorce días, mientras que las ratas control recibieron cantidades iguales de alimentos sin ańadir MSG. Las ratas fueron alimentadas con los productos adquiridos en Edo Feeds y Flour Mill Ltd, Ewu, Estado de Edo, y se les dio agua libremente. Las ratas fueron sacrificadas el día quince del experimento. El colliculus inferior fue cuidadosamente disecado y rápidamente fijado en 10% formal de solución salina para el estudio histológico convencional con de H&E.
Los hallazgos histológicos observados con H&E indicaron que las secciones del colliculus inferior de las ratas tratadas presentaban algunos cambios degenerativos celulares, hipertrofia celular, y vacuolas autofágicas, con algunas vacuolizaciones intercelulares que aparecen en el estroma, y cierto grado de hipertrofia neuronal en comparación con las secciones de control.
Estos resultados indican que el consumo de MSG puede tener un efecto perjudicial en las neuronas del colliculus inferior (IC). MSG probablemente puede tener efectos adversos sobre la sensibilidad auditiva y sobre las células nerviosas del colliculus inferior de ratas Wistar adultas. Se recomienda llevar a cabo nuevos estudios encaminados a corroborar estas observaciones.
Palabras Clave: Glutamato monosódico, colliculus inferior, vacuolizaciones intercelulares, ratas Wistar.
INTRODUCTION
Most food additives act either as preservatives or enhancer of palatability. One of such food additive is monosodium glutamate (MSG) and it is sold in most open markets and stores in Nigeria as "Ajinomoto" marketed by West African Seasoning Company Limited. Various environmental chemicals, industrial pollutants and food additives have been implicated as causing harmful effects1.
The safety of MSG's usage has generated much controversy locally and globally2. In Nigeria, most communities and individuals often use MSG as a bleaching agent for the removal of stains from clothes. There is a growing apprehension that its bleaching properties could be harmful or injurious to the body, or worse still inducing terminal diseases in consumers when ingested as a flavor enhancer in food. Despite evidence of negative consumer response to MSG, reputable international organizations and nutritionist have continued to endorse MSG, reiterating that it has no adverse reactions in humans. Notably of such is the Directorate and Regulatory Affairs of Food and Drug Administration and Control (FDA&C) in Nigeria, now NAFDAC has also expressed the view that MSG is not injurious to health3.
MSG improves the palatability of meals and thus influences the appetite centre positively with it resultant increase in body weight4. Though MSG improves taste stimulation and enhances appetite, reports indicate that it is toxic to human and experimental animals5. MSG has a toxic effect on the testis by causing a significant oligozoospermia and increase abnormal sperm morphology in a dose-dependent fashion in male Wistar rats6. It has been implicated in male infertility by causing testicular hemorrhage, degeneration and alteration of sperm cell population and morphology7.
It has been reported that MSG has neurotoxic effects resulting in brain cell damage, retinal degeneration, endocrine disorder and some pathological conditions such as addiction, stroke, epilepsy, brain trauma, neuropathic pain, schizophrenia, anxiety, depression, Parkinson's disease, Alzheimer's disease, Huntington's disease, and amyotrophic lateral sclerosis8. It cannot be stated that MSG is the cause of such varied conditions as epilepsy and Alzheimer's disease, although there may be concerns of its involvement in its etiology.
The inferior colliculus is an important relay centre in the acoustic (auditory) pathway. It receives fibres of the lateral lemniscus arising in the superior olivary complex. Each colliculus receives auditory impulses from both ears. These impulses are relayed to the medial geniculate body, through the inferior brachium and from there to the auditory area of the cerebral cortex. The inferior colliculus can influence motor neurons in the spinal cord and brainstem through the superior colliculus and the tectospinal and tectotegmental tracts9.
Since it has been reported that MSG has a neurotoxic effect, it is worthwhile to investigate its effects on the inferior colliculus. This work is carried out to investigate some probable histological effects of MSG on the IC, being an important relay centre in the acoustic (auditory) pathway and sub cortical relay structure for hearing in adult Wistar rats.
MATERIALS AND METHODS
ANIMALS:
Twenty four (24) adult Wistar rats of both sexes with average weight of 185g were randomly assigned into three groups A, B and C of (n=8) in each group. Groups A and B of (n=16) serves as treatments groups while Group C (n=8) is the control. The rats were obtained and maintained in the Animal Holdings of the Department of Anatomy, School of Basic Medical Sciences, University of Benin, Benin city, Nigeria.
They were fed with growers' mash obtained from Edo feed and flour mill limited, Ewu, Edo state) and given water liberally. The rats gained maximum acclimatization before actual commencement of the experiment. The Monosodium glutamate (3g/ sachet containing 99+% of MSG) was obtained from Kersmond grocery stores, Uselu, Benin City.
MONOSODIUM GLUTAMATE ADMINISTRATION:
The rats in the treatment groups (A and B) were given 3g and 6g of MSG, thoroughly mixed with the growers' mash, respectively. The control © group received equal amount of feeds (growers' mash) without MSG added for fourteen days. The rats were sacrificed on the fifteenth day of the experiment. The inferior colliculus was quickly dissected out and fixed in 10% formal saline for routine histological techniques. The 3g and 6g MSG doses were chosen and extrapolated in this experiment based on the indiscriminate use here in Nigeria due to its palatability. The two doses were thoroughly mixed with fixed amount of feeds (550g) in each group, daily.
HISTOLOGICAL STUDY:
The tissue were dehydrated in an ascending grade of alcohol (ethanol), cleared in xylene and embedded in paraffin wax. Serial sections of 7 microns thick were obtained using a rotatory microtome. The deparaffinized sections were stained routinely with haematoxyline and eosin. Photomicrographs of the desired sections were made for further observations.
RESULTS
The control sections of the inferior colliculus showed normal histological features with the neurons appearing distinct and the glial cells normal without vacuolations in the stroma (Figure 1).
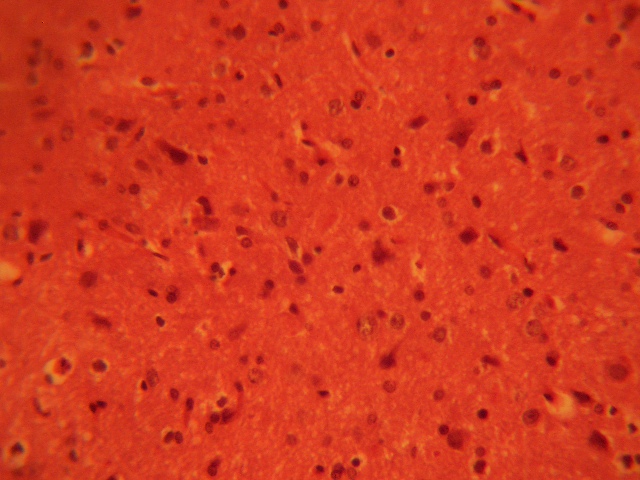
FIGURE 1: Control section of the inferior colliculus. (Original magnification x 400)
The treatment sections of the inferior colliculus showed some varying degree of reduced cellular population based on its sparse distribution, degenerative changes, cellular hypertrophy, and intercellular vacuolations appearing in the stroma; more marked in the treatment sections receiving 6g of MSG (Figure 2 & 3).
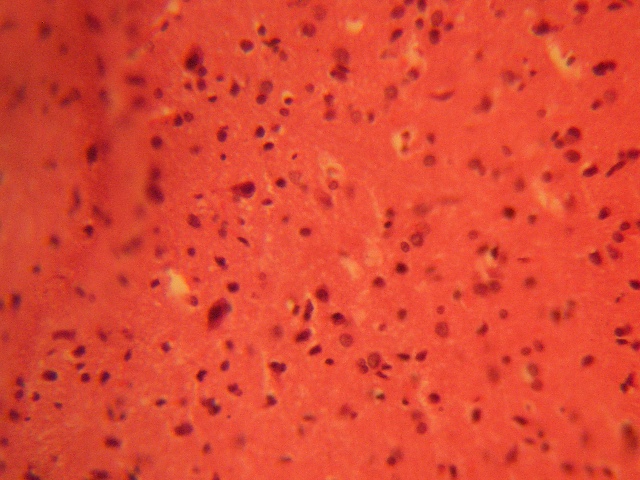
FIGURE 2: Treatment section of the inferior colliculus (3g MSG) (Original magnification x 400)
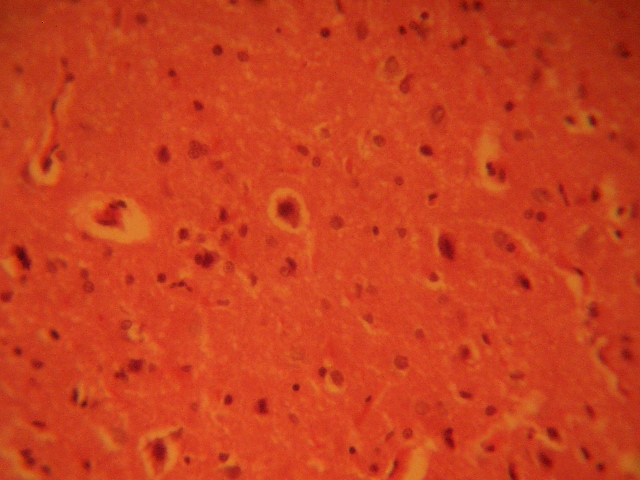
FIGURE 3: Treatment section of the inferior colliculus (6g MSG) (Original magnification x 400)
DISCUSSION
The results (H & E) revealed that with increasing dose of monosodium glutamate consumption caused cellular degenerative changes, cellular hypertrophy and autophagic with intercellular vacuolations appearing in the stroma of the treatment groups compared to the control section of the inferior colliculus. Neuronal degeneration has been reported to result in cell death, which is of two types, namely apoptotic and necrotic cell death. These two types differ morphologically and biochemically10. Pathological or accidental cell death is regarded as necrotic and could result from extrinsic insults to the cell such as osmotic, thermal, toxic and traumatic effects11. It was reported that cell death in response to neurotoxins might trigger an apoptotic death pathway within brain cells12. Cell death in response to neurotoxins occurs as a controlled event involving a genetic programme in which caspase enzymes are activated12.
The process of cellular necrosis involves disruption of the membranes structural and functional integrity. Cellular necrosis is not induced by stimuli intrinsic to the cells as in programmed cell death (PCD), but by an abrupt environmental perturbation and departure from the normal physiological conditions13. There is the need to further investigate the actual mechanism by which MSG induced neuronal degeneration in the inferior colliculus of adult Wistar rat in this study.
Extensive cell death in the central nervous system is present in all neurodegenerative diseases12. The type of nerve cell loss and the particular part of the brain affected dictate the symptoms associated with an individual disease12. In this study MSG may have acted as toxin to the cells of the lateral geniculate body, affecting their cellular integrity and causing defect in membrane permeability and cell volume homeostasis.
In cellular necrosis, the rate of progression depends on the severity of the environmental insults. The greater the severity of the insults the more rapid the progression of neuronal injury13. The principle holds true for toxicological insult to the brain and other organs14. The prime candidates for inducing the massive cell destruction observed in neurodegeneration are neurotoxins12. These may be substances present in small amounts in the environment, or even naturally occurring chemicals such as glutamate used by the brain as transmitter's substances12. The latter when present at a critical level can be toxic to the brain cells they normally excite12. It is inferred from this results that prolonged and high dose of MSG resulted in increased toxic effects on the IC. In the same way, it has been reported that MSG consumption in food resulted in the cellular degenerative changes and vacuolations appearing in the stroma with some autophagic vacuoles in the medial geniculate body of adult Wistar rats15.
The vacuolations observed in the stroma of the inferior colliculus in this experiment may be due to MSG interference, since it has been reported that MSG may be neurotoxic to cerebella development in chick16, and also produces neuronal degeneration in several brain regions when administered in neonatal rats17. The cellular hypertrophy observed in this experiment may be due to the cytotoxic effects of MSG on the inferior colliculus. This obviously will affect the normal propagation of impulses in this intracranial auditory relay centre. Normal conduction of impulse involves the propagation of action potential along nerve fibers in the form of wave of activity that constitutes the nerve impulse18.
CONCLUSION AND RECOMMENDATION
The results obtained in this study revealed that monosodium glutamate consumption could affect the histology of the inferior colliculus. The nerve cells of the treated sections of the inferior colliculus showed some cellular degenerative changes, autophagic vacuoles and neuronal hypertrophy. With this result it is probable that the functions of the inferior colliculus as an intracranial auditory relay centre may be adversely affected. It is recommended that further studies be carried out to corroborate these findings.
REFERENCES
1.- Moore KL. Congenital malformations due to environmental; Developing Humans. W.B. Saunders co. Ltd. Philadelphia. 2003; 2nd ed. 173-183.
2.- Biodun D, Biodun A. A spice or poison?. Is monosodium glutamate safe for human consumption? National concord 1993; 4th Jan. 5.
3.- Okwuraiwe PE. The role of food and Drug Administration and control (FDA&C) in ensuring the safety of food and food Ingredients: A symposium held at Sheraton Hotel, Lagos. 1st Sept.1992: 6-15.
4.- Rogers PP, Blundell JE. Umani and appetite: Effects of monosodium glutamate on hunger and food intake in human subjects. Phisiol Behav. 1990; 486:801-804.
5.- Belluardo M, Mudo G, Bindoni M. Effect of early destruction of the mouse arcuate nucleus by MSG on age dependent natural killer activity. Brain Res. 1990; 534:225-333
6.- Onakewhor JUE, Oforofuo IAO, Singh SP. Chronic administration of monosodium glutamate Induces Oligozoospermia and glycogen accumulation in Wister rat testes. Africa J Reprod Health 1998; 2: 190-197.
7.- Oforofuo IAO, Onakewhor JUE, Idaewor PE. The effect of chronic admin. Of MSG on the histology of the Adult wister rat testes: Bioscience Research Communications. 1997:9, No. 2
8.- Samuels A. The Toxicity/Safety of MSG; A study in suppression of information. Accountability in Research 1999; 6: 259-310.
9.- Singh I. Textbook of Human Neuroanatomy, 6th ed. Jaypee Brothers Medical Publishers (P) Ltd. 2002: 126-129.
10.- Wyllie AH. Glucocorticoid-induced thymocyte apoptosis is associated with endogenous endonuclease activation. Nature: London 1980; 284:555-556.
11.- Farber J L, Chein K R, Mittnacht S. The pathogenesis of Irreversible cell injury in ischemia; American Journal of Pathology 1981; 102:271-281.
12.- Waters CM, Wakinshaw G, Moser B, Mitchell IJ. Death of neurons in the neonatal rodent globus pallidus occurs as a mechanism of apoptosis. Neuroscience. 1994; 63: 881-894.
13.- Ito U, Sparts M, Walker JR, Warzo I. Experimental Cerebral Ischemia in Magolian Gerbils(1). Light microscope observations. Acta Neuropathology. USA. 2003; 32:209-223.
14.- Martins LJ, Al-Abdulla NA, Kirsh JR, Sieber FE, Portera-Cailliau C. Neurodegeneration in excitotoxicity, global cerebral ischaemia and target deprivation: A perspective on the contributions of apoptosis and necrosis. Brain Res Bull. 1978; 46: 281-309.
15.- Eweka AO, Adjene JO. Histological studies of the effects of monosodium glutamate on the medial geniculate body of adult Wister rat. Electron J Biomed 2007;2:9-13.
16.- Espinar A, Garcia-Oliver A et al. Neuroprotection by melatonin from glutamate-induced exocitoxicity during development of the cerebellum in the chick embryo. J Pineal Res 2000; 2: 818.
17.- Urena-Guerrero ME, Lopez-perez SJ, BeasZaratel B. Neonatal Monosodium glutamate treatment modified glutamic acid decarboxylase activity during rat brain postnatal development: Neurochem. Int. 2003; 42: 269-276.
18.- Gispen WH. Molecular and functional neurobiology: Elsevier Publishing Company, USA 1976; 102-104.
Corresponding author
Dr. A. O. Eweka
Department of Anatomy. School of Basic Medical Sciences. College of Medical Sciences, University of Benin.
Benin City. Edo State. Nigeria.
andreweweka @ yahoo.com
Received January 31, 2008
Published October 4, 2008
