Indice del volumen Volume index
Comité Editorial Editorial Board
Comité Científico Scientific Committee
- Group IIa, 10 mg/kg b.w extract of Jatropha gossypifolia
- Group IIIa, 20 mg/kg b.w extract of Jatropha gossypifolia
- Group IVa, 30 mg/kg b.w extract of Jatropha gossypifolia
- Group IIb, 10 mg/kg b.w extract plus 10 mg/kg b.w Prednisolone
- Group IIIb, 20 mg/kg b.w extract plus 10 mg/kg b.w Prednisolone
- Group IVb, 30 mg/kg b.w extract plus 10 mg/kg b.w Prednisolone
- Each group and subgroup were further divided into three classes A, B and C as below:
EFFECTS OF ADMINISTRATION OF ETHANOLIC ROOT EXTRACT OF JATROPHA GOSSYPIFOLIA AND PREDNISOLONE ON THE KIDNEYS OF WISTAR RATS
Medubi L.J.1, Ukwenya V.O.2, Aderinto O.T.3, Makanjuola V.O.4 , Ojo O.A.5, Bamidele O.5, Ajao M.S.1.
1Departments of Anatomy, Faculty of Basic Medical Sciences, College of Health Sciences, University of Ilorin, Kwara State and
4 Bingham University, Nassarawa.
2Departments of Anatomy and 5Physiology, Faculty of Basic Medical Sciences, College of Health Sciences, Bowen University, Iwo, Osun State.
3Department of Physiology, Faculty of Basic Medical Sciences, College of Health Sciences, University of Lagos, Lagos.
Nigeria
victorwyn @ yahoo.com
Rev Electron Biomed / Electron J Biomed 2010;2:41-48
Comment of the reviewer Prof. Pilar Muńiz Rodríguez PhD. Department of Biochemist and Molecular Biology. Faculty of Sciences. Universidad de Burgos. Spain.
Comment of the reviewer Dra. Silvia Albillos, PhD. Institute of Biotechnology (INBIOTEC). León. Spain.
ABSTRACT
The effect of oral administration of ethanolic root extract of Jatropha gossypifolia and prednisolone on the kidney histology and renal function of albino rats was studied to assess the safety and toxicity of the plant as an herbal remedy.
The rats were divided into four groups I, II, III and IV. Group I served as control and was given feed and water only. Group II, III, and IV were subdivided into Group IIa, IIb, IIIa, IIIb, IVa and IVb. Groups IIa, IIIa, and IVa received 10 mg, 20 mg and 30 mg/kg b.w of the extract while Group IIb, IIIb and IVb received 10 mg ,20 mg and 30 mg/kg b.w of the extract respectively plus 10 mg/kg b.w of prednisolone per day. The animals were sacrificed on day 7, 10 and 14 and their kidneys harvested and processed for histological studies. Their blood was also collected for serum urea measurement.
Photomicrographs of the histological sections of Groups II, III and IV rats revealed changes compared to the control group and serum urea levels were significantly higher in these groups. Histological changes observed are consistent with glomerulonephritis and include increased urinary (Bowman's) space, shrinkage and distortion of the glomerular tuft as well as scarring of the glomeruli. Changes appear to be both dosage and time dependent and the administration of prednisolone as an adjunct did not exert any ameliorative effect.
We conclude that ethanolic root extract of Jatropha gossypifolia is toxic to the kidney and causes increased urea retention in the blood.
KEYWORDS: Jatropha gossypifolia, serum urea, Prednisolone, glomerulonephritis, urinary space.
RESUMEN : EFECTO DE LA ADMINISTRACIÓN DE EXTRACTO ENÓLICO DE LA RAÍZ DE JATROPHA GOSSYPIFOLIA Y PREDNISOLONA EN EL RIŃON DE RATAS WISTAR
Ha sido estudiado el efecto de la administración oral de extracto etanólico de la raíz de gossypifolia Jatropha y prednisolona en la histología renal y la función renal de ratas wistar, para evaluar la seguridad y la toxicidad de la planta como un remedio herbal.
Las ratas fueron divididas en cuatro grupos I, II, III y IV. El grupo I fué utilizado como control y recibió solamente alimento y agua. Los grupos II, III y IV fueron subdivididos en grupos IIa, IIb, IIIa, IIIb, IVa y IVb. Los grupos IIa, IIIa y IVa recibieron 10 mg, 20 mg y 30 mg /kg de peso corporal de extracto mientras que el grupo IIb, IIIb y IVb recibieron respectivamente 10 mg, 20 mg y 30 mg /kg de peso corporal de extracto, más 10 mg/kg peso corporal de prednisolona por día. Los animales se sacrificaron en los días 7, 10 y 14. Los rińones fueron extraídos y procesados para estudios histológicos. Se recogió también sangre para determinación de urea sérica.
Las microfotografías de las secciones histológicas de los grupos II, III y IV ratas revelaron cambios en comparación con el grupo control, y los niveles séricos de urea fueron significativamente mayores en estos grupos. Los cambios histológicos observados son coherentes con glomerulonefritis e incluyen aumento del espacio de Bowman, la retracción y distorsión del penacho glomerular, así como cicatrices glomérulares. Los cambios parecen ser dependientes de la dosis y el tiempo y la administración de prednisolona como un complemento no ejerce ningún efecto protectos.
Se concluye que la raíz del extracto etanólico de gossypifolia Jatropha es tóxica para el rińón y provoca retención de urea en sangre.
PALABRAS CLAVES: Gossypifolia Jatropha. Urea sérica. Prednisolona. Glomerulonefritis. Espacio urinario.
INTRODUCTION
Jatropha gossypifolia belongs to the family Euphorbiaceae and the order Genanialae. The common names include Belly ache bush, cotton leaf, physic nut, sibigua and tuatua. The Yorubas of Western Nigeria call it Lapalapa pupa1 or Botuje-pupa2.
Jatropha gossypifolia is an herbaceous plant that rarely grows more than 1 meter tall. Literature about the ornamental and traditional uses of Jatropha gossypifolia abound.The leaf decoction of Jatropha gossypifolia is used for bathing wounds and the stem sap used to stop bleeding and itching of cuts and scratches3. Oduola et al4 reported that the raw extract of the leaf of Jatropha gossypifolia has anticoagulant activity and it was opined that if the active chemicals are isolated and purified the leaf extract could be used for therapeutic control of thrombosis.In some parts of Nigeria the root decoction in addition to salt is used to treat syphilis, general illness and gonorrhea5.
Since the Jatropha species bear some resemblance to each other, a non-discerning individual could call them by the same name and use them indiscriminately. This has been reported to result in unintentional poisonings6-7.
Prednisolone is the active metabolite of prednisone, a glucocorticoid drug with potent immunosuppressant and anti-inflammatory effects8. It exerts its anti-inflammatory effects by preventing the agglutination and mobilization of neutrophils to inflamed areas9, by reducing the synthesis of arachidonic acid by phospholipase A2 by inhibiting the expression of COX-210 and by exerting anti-oxidant effects11.
Sjoden and Lindgren12 reported the efficacy of prednisolone in treating kidney toxity induced by vitamin D overdose. The drug has also been reported to reverse acute interstitial nephritis induced by cotrimoxazole 8.
Due to the indiscriminate and increasing use of plants for herbal remedy in the African sub region, it is important that scientific experimentation be carried out to ascertain the safety and possible side effects of the various parts of these plants on sensitive organs of the body like the kidney. This study was aimed at investigating the effect of this popular member of the Jatropha species on the kidney.
MATERIALS AND METHODS
Plant Material
The root of Jatropha gossypifolia was collected at Akure, Ondo State, Nigeria and was authentically identified at the herbarium of the Department of plant Biology, University of Ilorin.
Extract Preparation
The root bark was removed and sun-dried for seven days after which it was oven-dried at 50 oC for 24 hours to constant weight. It was ground into fine powder and weighed 520g weighed. The powder was soaked in 10L of 70% ethanol for 24 hours and extracted with Soxhlet. The extract was concentrated using Rotary Evaporator (Laborato 4000, China). A final weight of 35.70g of the ethanolic extract was obtained representing 6.87% yield of the dry powder. A standard concentration of 10mg/ml was prepared by dissolving 10g in 1000mL of distilled water.
Animal Material
One hundred and five albino rats (Rattus novergicus) weighing between 100-190g were procured from the animal house,Department of Biochemistry,University of Ilorin, Nigeria were acclimatized for two weeks at 27°c and 12 hours light/dark cycle. They were fed with grower mash (Bendel Feeds and Flour Limited, Ilorin) with clean water ad libitum.
Experimental Design
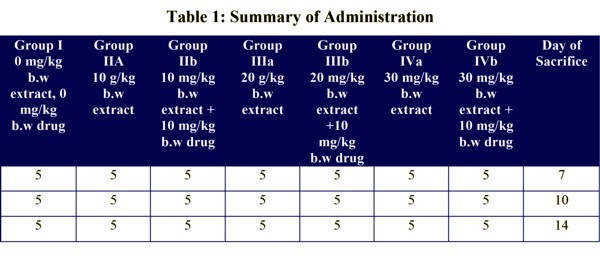
The rats were divided into groups I, II, III and IV respectively. Group I served as the Control while Groups II, III and IV were subdivided into two groups each, which are IIa, IIb, IIIa, IIIb, IVa and IVb.
In addition to the normal feed and water the groups received the following extract/drug by oral intubation:
All procedures involving the use of animals in this study complied to the guiding principles for research involving animals as recommended by the declaration of Helsinki and the Guiding Principles in the care and use of animals13.
Blood collection
Twenty-four hours after the last doses were administered for each of the Classes, the animals were anaesthetised with chloroform vapour and blood samples were collected by left cardiac puncture in the carotid arteries. The blood samples were collected into clean centrifuge tubes and the serum urea concentration, which is indicative of renal function, was determined by the method described by Kassirer J.P 14.
It should be noted that the animals were sacrificed in subgroups as indicated in Table 1.
Tissue preparation for Histology
The kidneys were carefully dissected out, trimmed of all fat and blotted dry to remove blood. Their weight was noted and fixed in 10% formol-saline. The fixed tissues were transferred to a graded series of ethanol and then cleared in xylene. Once cleared, the tissues were infiltrated in molten paraffin wax in the oven at 58oC. Serial sections of 5µm thick were obtained from a solid block of tissue, fixed on clean slides stained with haematoxylin and eosin stains, and examined with the light microscope.
Statistical Analysis
Data were expressed as means ± standard deviation (SD). Statistical analyses were performed using one-way Analysis of variance (ANOVA) test and the Scheffe's post-hoc test. Unless mentioned otherwise, a p value < 0.05 was regarded as statistically significant.
RESULTS
Serum Urea Concentration
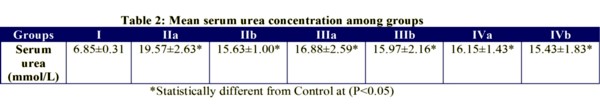
Serum concentration was significantly increased (p<0.05) in each of the treated groups compared with the control. The serum urea levels (all values in mmol/L) were 6.85±0.31 for control rats, 19.57±2.63 and 16.88±2.59 respectively in those administered 10 mg and 20 mg of the extract; and 16.15±1.43 in those given 30 mg of the extract. The mean serum urea concentration was significantly lower(p<0.05) in Group IIb rats which received 10 mg of Prednisolone and 10 mg of the extract compared with Group IIa animals which received just the extract. However, there was no statistical significant difference between Groups IIIa and IIIb as well as between Groups IVa and IVb (Table 2).
The effect of the extract on serum urea concentration appears to be time-dependent with the serum urea concentration and significantly different on days 7,10 and 14 respectively. The mean value was highest on day 10, lowest on day 7 and medium on day 14 (Table 3).
Histology
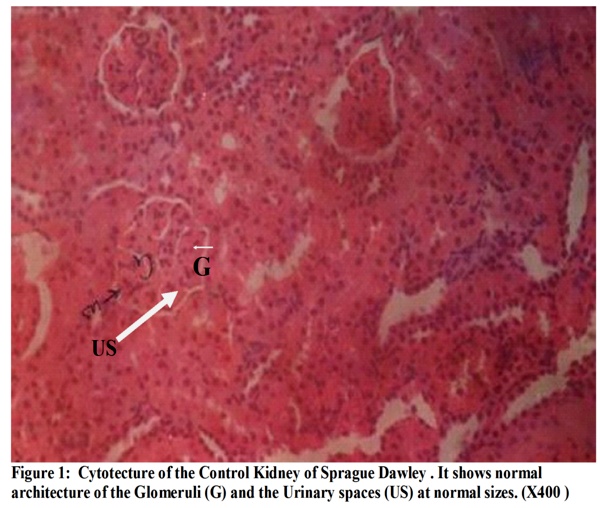
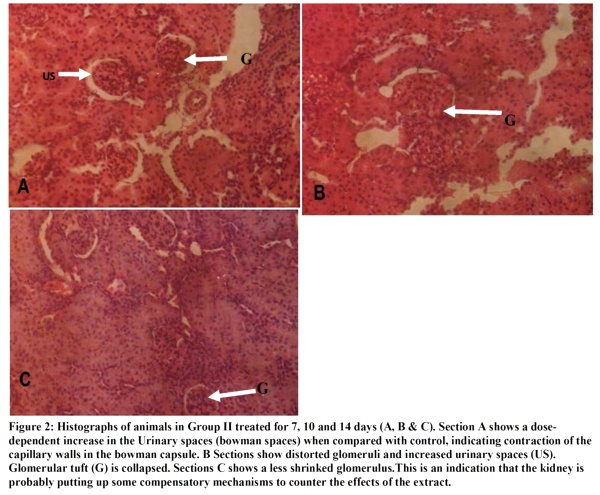
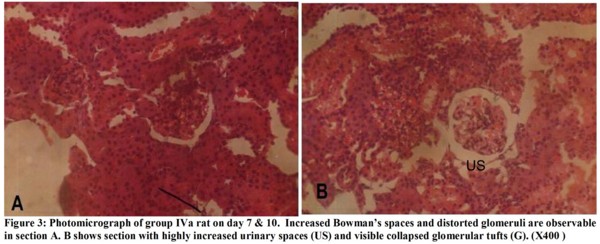
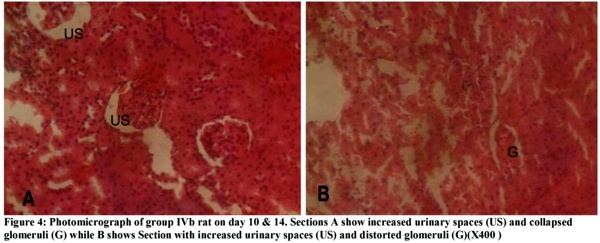
The micrographs (Figures 1-4) revealed changes associated with the glomerular tuft and Bowman's spaces. There was shrinkage and distortion of the glomerular tufts as well as increase in the Bowman's spaces. Also observable are scarring of the glomerulus and interstitial fibrosis, which are consistent with glomerulonephritis. These changes appear to be both dosage and time-dependent as the distortion of the glomeruli is highest in rats administered 30 mg of the extract, medium in those given 20mg and least in those that received 10mg. Animals that were sacrificed on day 14 have their glomeruli more distorted than those of day 10 while animals sacrificed on day 7 were the least affected.
DISCUSSION
The kidneys are the organs responsible for the excretion of toxic substances from the body. Their structural integrity and functional efficiency can be adversely affected when noxious substances have to be cleared from the body.
The glomerular distortion and increased urinary spaces seen in the kidneys of experimental animals may be due to the spasmolytic and vasodilatory effects of the extract. These conditions are associated with prolonged reduction in the percentage of cardiac output to the kidneys, which causes reduced glomerular pressure and collapse of the glomerular capillaries. Dilation of vessels all over the body for a prolonged time can cause cardiac depression, which can eventually lead to reduced glomerular capillary pressure and increased urinary spaces. The mesengial cells of the kidneys are contractile and modify the diameter of the glomerular capillaries in response to vasoactive substances. This agrees with Ojewole and Odebiyi5 findings that teramethylpyrazine(TMPZ), isolated from stems of Jatropha specie, possesses specific spasmolytic and vasodilator activity. General reduction in body fluid and extracellular fluid caused by haemorrhage, vomiting and diarrhoea can cause glomerular hypotension, which can also result in glomerular shrinkage, distortion and increased urinary spaces.
In addition to the histological findings, serum urea concentration was measured to ascertain the functional integrity of the kidney during the period of administration of the extract and prednisolone. Urea is the end product of protein metabolism. High serum urea concentration occurs when the amount excreted falls below that formed by metabolism. Kumar et al 15 reported that elevation of of blood urea nitrogen and creatinine levels is largely related to decreased glomerular filtration rate. The elevated serum urea levels seen in this study thus signifies that the plant extract impeded kidney excretory function.
Blood urea level can also be raised in conditions associated with depletion of extracellular fluid and plasma volume. Bleeding into the GIT and any form of internal hemorrhage also causes increased urea concentration. Thus the elevated serum urea level recorded might be due to the anti-coagulant effects of the plant extract in vivo. This agrees with the findings of Oduola et al4 that the sap of Jatropha gossypifolia is an anti-coagulant. It might also be due to the action of TMPZ, an amide alkaloid present in the root of the plant. TMPZ is known to cause cardiovascular and respiratory depression, which in turn can cause increased urea concentration.
It is noteworthy that the mean serum urea concentration was significantly lower in Group IIb animals which received 10 mg of prednisolone and 10 mg of the extract, compared with Group IIa animals which received only the extract. This is in consonance with the fact that prednisolone is a mineralocorticoids and in this case might have facilitated the rate of excretion of the Jatropha root metabolites. However, there was no statistical significant difference between the urea serum levels of Group IIIa and IIIb as well as between Group IVa and IVb. This may suggest that 10 mg of the drug is not potent against the effects of 20 mg and 30 mg of the extract, but may exert some effects at lower doses. Furthermore, the histological sections from the kidneys of groups IIIb and IVb rats do not indicate that prednisolone exerted any ameliorative effects.
The effect of the extract on serum urea concentration is also time-dependent: lowest on day 7, highest on day 10 and medium on day 14.This is probably due to some compensatory mechanism put in place by the kidney to reduce the effect of the extract.
From the foregoing it was observed that that ethanolic extract of Jatropha gossypifolia is toxic to the kidneys at the dose used in this work and that prednisolone exerted very limited protective effects. We therefore suggest that more research be carried out on eliciting the mechanism of action of Jatropha gossypifolia.
REFERENCES
-
1. Odebiyi OO, Sofowora EA. Phytochemical screening of Nigerian medicinal plants II. Lloydia. 1978;41(3):234-46.
2. Soladoye MO, Sonibare MA, Nach AO Alabi DA. Indiginuous angiosperm biodiversity of Olabisi Onabanjo University, Permanent Site. Afr J Biotechnol. 2005;4:562-564.
3. Horsten SF, van den Berg AJ, Kettenes-van den Bosch JJ, Leeflang BR, Labadie RP. Cyclogossine A: a novel cyclic heptapeptide isolated from the latex of Jatropha gossypifolia. Planta Med. 1996;62:46-50.
4. Oduola T, Avwioro OG, Ayanniyi TB. Suitability of the leaf extract of Jatropha gossypifolia as an anticoagulant for biochemical and haematological analyses. Afr J Biotechnol. 2005;4:677-681.
5. Ojewole JAO, Odebiyi OO. Neuromuscular and Cardiovascular Actions of Tetramethylpyrazine from the Stem of Jatropha podagrica. Planta Med. 1980; 38:332-338.
6. Abdu-Aguye I, Sannusi A, Alafiya-Tayo RA, Bhusnurmath SR. Acute toxicity studies with Jatropha curcas L. Hum Toxicol. 1986;5:269-274.
7. Consroe PF, Glow DE. Clinical toxicology of desert potato: Two case reports of acute Jatropha root ingestion. Arizona Med. 1975;23:475-477.
8. Mozaffari A, Rashidi I. Investigation of protective effect of prednisolone in nephropathy induced by co-trimoxazole in rat. Pak J Med Sci 2007;23:847-850
9. Mann BS, Chung KF. Blood neutrophil activation markers in severe asthma: lack of inhibition by prednisolone therapy. Respir Res. 2006;7:59.
10. Trevor AJ, Katzung BG, Masters SB. Katzung and Trevor's Pharmacology: examination and board review. 7th Edition, 2005; 176.
11. Yazar E, Konyalioglu S, Col R, Birdane YO, Bas AL, Elmas M. Effects of vitamin E and prednisolone on some oxidative stress markers in endotoxemic rabbits. Revue Méd. Vét.,2004; 155, 11, 538-542.
12. Sjöden G, Lindgren U. The effect of prednisolone on kidney calcification in vitamin D-treated rats. Calcif Tissue Int. 1985;37:613-616.
13. World Medical Association; American Physiological Society. Guiding principles for research involving animals and human beings. Am J Physiol Regul Integr Comp Physiol. 2002;283:R281-R283.
14. Kassirer JP. Clinical evaluation of kidney function--glomerular function. N Engl J Med. 1971;285:385-389.
15. Kumar V, Abbas AK, Fausto N, Mitchel R. Robbins Basic Pathology. 8th Edition, 2007; 542.
Correspondence:
Ukwenya Victor Okoliko
Department of Anatomy, Faculty of Basic Medical Sciences,
College of Health Sciences,
Bowen University, Iwo
Osun State. Nigeria
Email: victorwyn @ yahoo.com