Indice del volumen Volume index
Comité Editorial Editorial Board
Comité Científico Scientific Committee
CHORIONIC VILLI IN THE CHRONIC ENDOMETRITIS.
Olivar C Castejon S. Bsc. Msc.
Magister Scientiarum, Full Professor in Biology.
Laboratory of Electron Microscopy. Center for Research and analysis assistancel teaching of the nucleus Aragua(Ciadana).
Faculty of Health Sciences. University of Carabobo. Aragua State. Maracay. Venezuela.
Email: olivar.ciadanauc @ gmail.com
Rev Electron Biomed / Electron J Biomed 2018;3:23-35.
SUMMARY
Objective: The aim of this study was to examine the structure of the placental villi in the chronic endometritis at 8 weeks of pregnancy without another antecedent of illness and evaluated with light microscopy.
Study design: Placenta study and placenta normal were compared. Placenta study proceeds of pacient of 28 years old with problems of infertility and medical history of recurrent abortion and dysfunctional uterine bleeding.
Results: The decidual region showed an infiltration of leucocytes polimorphonuclears with plasma cells. The villi showed anaplasic feature, increased growth and multiplication of citotrophoblast cells associate with giant multinuclear cells, intervillous thrombosis, immaturity, debris of tissue death and degenerated villi associate to endometrium in degeneration.
Conclusion: These results indicate the vulnerability of the trophoblast during infection increasing the mechanisms that produce the abortion.
KEY WORDS: Chorionic villi endometritis abortion
INTRODUCTION:
Endometritis is an infectious and inflammatory disorder of the endometrium. Chronic endometritis (CE) is characterized by endometrial superficial edematous change, high stromal cell density; dissociated maturation between epithelium and stroma, and infiltration of endometrial stromal plasmocytes cells (ESPCs)1. The presence of multiple ESPCs is the most frecuent findings in this pathology2. This long-neglected gynecologic disease, with subtle symtoms as pelvic discomfort, spotting, leucorrhea, often unnoticed by patients, being a benign disease, not has been described as associated with placental villi and histopathologic examinations appear not to be favored by gynecologic practice3.
The microorganisms in endometrium with chronic endometritis are: Streptococcus species, Escherichia coli, Enterococcus faecalis, Staphylococcus species, Mycoplasm/Ureaplasma species: Mycoplasma genitalium, Mycoplasma hominis, and Ureaplasma urealyticum, Proteus species, Klebsiella pneumonia, Psuedomonas aeruginosa, Gardnerella vaginalis,Chlamydia trachomatis,Neisseria gonorrhoeae, Coryne-bacterium and yeasts: Sacharomyces cerevisiae and candida species4. Mycobacterium tuberculosis has been mentioned as a microorganism causing a poorly developed caseating granuloma and surrounding lymphocyte infiltrates including ESPCs5. Altered proportions in anaerobic lactobacilli species, human immunodeficiency virus and cytomegalovirus also have been reported in CE. The histopatologic detection of multiple ESPCs in endometrial biopsy is of primary importance in the diagnosis of CE in current clinical practice3.
Plasmocytes typically have a large cell body, high nuclei/cytoplasm ratio, basophilic cytoplasm and nuclei with heterochromatin rearrangement as the "spoke wheel"6.The identification of glandular-stromal dyssynchrony and endometrial eosinophil infiltrates (cytoplasmic eosinophilic granules) on routine hematoxylin and eosin staining sections was proposed as a convenient screening tool to discover ESPCs but are not the absolute findings in CE7.
Histopathologic evaluation using immunohistochemistry for plasmocyte marker CD138 (Known as syndecan-1, a transmembrane type heparin sulfate proteoglycan) is currently the most reliable and time saving diagnostic method for CE8.
Strigent criteria have not yet been established for the evaluation of the ESPCs density in the endometrial biopsy specimen although the presence of multiple (two or more) ESPCs is a sine qua non for the confirmation of CE. There are some biases and variances in definitions of CE among the studies. While one study diagnosed CE with more of five CD138(+)ESPCs in at least one out of three sections levels in the endometrial biopsy specimens, others set the values for CE as one or more CD138(+)ESPCs in one macroscopic high-power field9.
Maternal infections caused by the microorganisms mentioned which can cross the placenta, may result in abortion or stillbirth. Pathogens can reach the deciduas only by dissemination in maternal cells, most likely leukocytes. If the defense mechanisms of the extravillous trophoblast are overcome, the infection may spread to the fetal blood, act as a nichus for maternal reinfection, and or cause trophoblastic death resulting in placental insufficiency or spontaneous abortion. Some pathogens may reach the fetus by traveling within maternal leukocytes on their natural way to the fetus10.The visualization of the placental villi and their interactions with the endometrium in this hostile ambient not have been observed and require to be investigated.
The aim of this study was to describe the histomorphology of the placental villi during this inflammatory process associated to endometritis.
MATERIAL AND METHODS:
Pacient of 28 years old who refers in her medical history intrauterine death to 8 weeks of gestation and abortion, with clinical antecedents of pelvic discomfort, spotting, leucorrhea, with anterior abortion, problems of infertility, dysfunctional uterine bleeding and endometrial sampling for histological investigation. Diagnosed with endometritis to the six weeks of the gestation. The infected woman pregnancy had knowledge of informed consent and approval by the ethical committee of the hospitalary institution for the realization of this investigation according to the Helsinky Declaration. One specimen of endometrial biopsy was taken in the hospitalary institution and sent to us for their microscopic study. No microbiologic investigation of the material was realized. Ten slides microscopic were prepared and stained with H&E for their observation. Histological diagnosis of chronic endometritis was based on the criteria previously described11, stromal edema, and hyperhemia. Chronic endometritis was defined as the presence of plasma cells in the endometrial stroma, including superficial stromal edema, increased stromal density, spindled stroma and polymorphic inflammatory cells1, 12.
RESULTS:
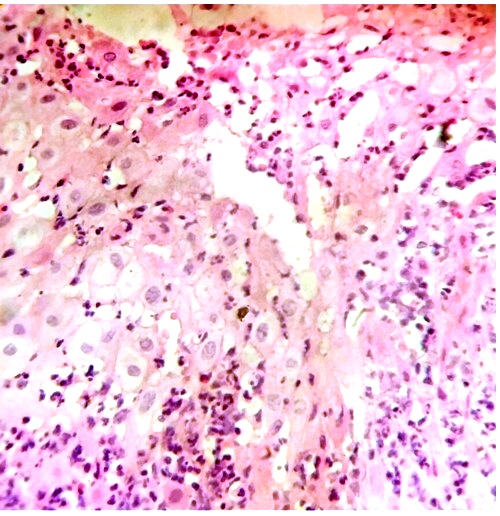
Observations realized in the deciduas permited to identify an infiltration of leukocytes polimorphonuclears which indicates an infectious and inflammatory disorder (Fig.1).
In this numerous poblation of leukocytes, plasma cells and eosinophil leukocytes were observed (Fig2).
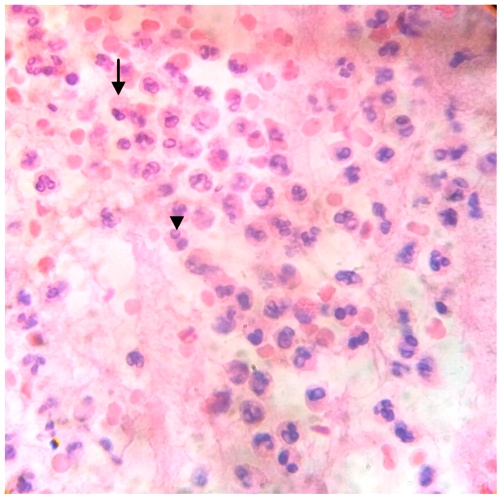
Fig.2. Plasma cells (arrow) and Eosinophil leucocytes (head of arrow) are seen in this section of degenerated tissue. H&E.400x
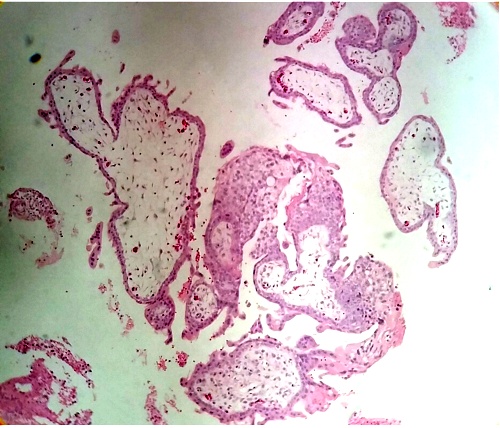
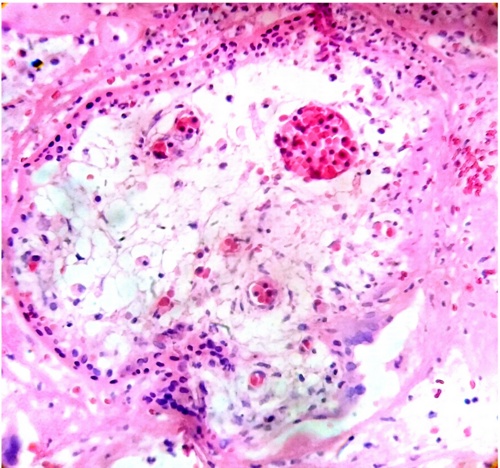
In some regions of the endometrium the placental villi appeared showing their anaplastic feature at level of the trophoblast (Fig.3).
An increased growth and multiplication of citotrophoblast cell associate with giant multinuclear cells was observed in the endometrial biopsy (Fig.4).
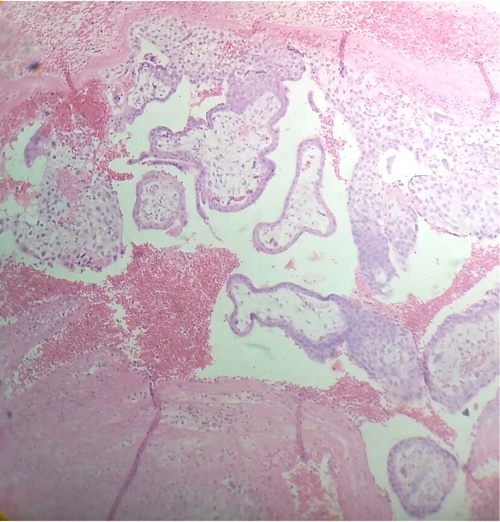
Thrombosis intervillous can to be noted in some regions of the endometrium (Fig.5).
Some villi shows their immaturity with stromal vessels that contain nucleated erythrocytes (Fig.6).
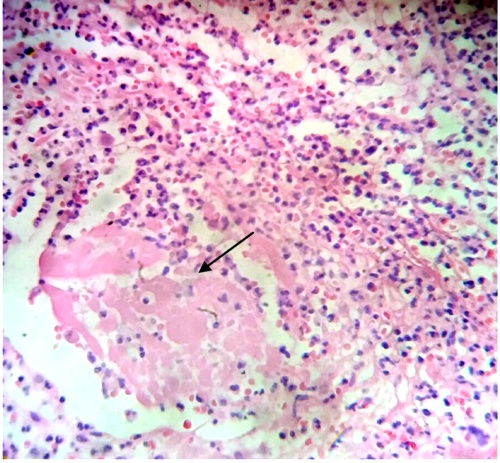
In the endometrial tissue debris of tissue death were observed (Fig.7).
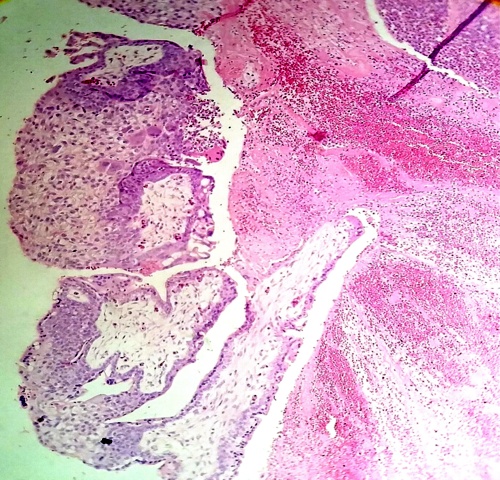
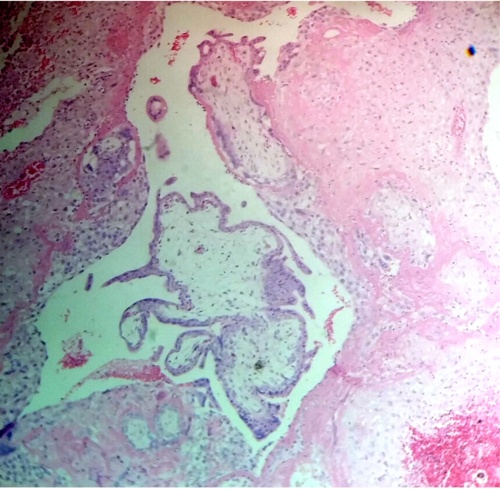
Chorionic villi is observed in conexion with an inflammed endometrium and in other regions they are as separating (Figs.8,9).
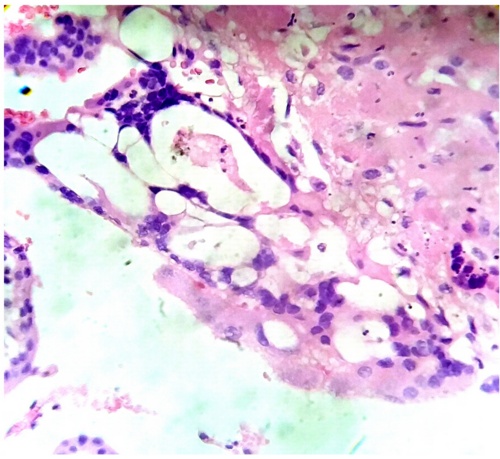
Fig.8. Both structures,placental villi and endometrium,are suffering a degenerative process. H&E.400x
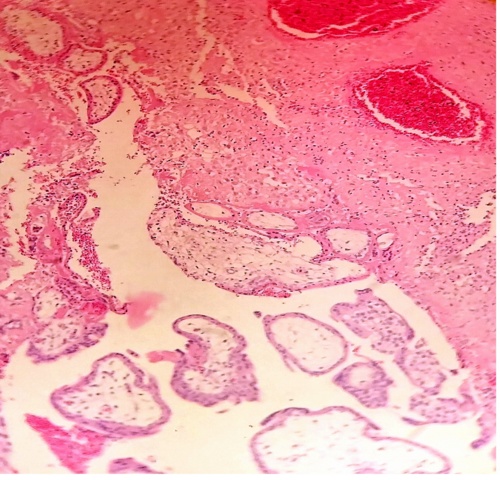
Fig.9. The villi which penetrated the endometrium have degenerated in their interior and has tendency to be separated. H&E.100x
Degenerated villi are seen associated to the endometrium which also suffers a process of degeneration (Fig.10).
DISCUSIÓN
The microorganisms of the Group B streptococci, Escherichia coli, Corynebacterium, Gardnerella vaginalis, Ureaplasma urealyticum, Micoplasma and Staphylococcus may cause chorioamnionitis during pregnancy and provoque repeated abortion. In our case an acute and chronic deciduitis in an immature placenta with chorioamnionitis inflamed the region where the placenta was inserted and the posterior necrosis of the zone makes a premature rupture13.
The identification of plasma cells and eosinophils cells in endometrial tissue could to be seen with light microscopy in this material obtained of abortion and the presence of polimorphonuclear leukocytes (PMNLs) was also observed in the villi.
The ascendant infection inflamed the membranes and the amniotic cells sinthetized prostaglandins that increased the delivery and the PMNLs secreted elastases and colagenases that initiated the premature rupture14, 15.
In their anaplastic feature the prolongations of the trophoblast intended to penetrate the endometrium and sections of villi were seen associate to the stromal region where suffered degenerative changes. This proliferative extent of trophoblast has been examined in specimens from spontaneous abortions that were seen in trisomy 7 and 1516. The diminished blood flow into the intervillous space at an early gestational age can to produce thrombosis intervillous which is of importance in causing spontaneous abortion13.
The observation of nucleated erythrocytes in these immature intermediate villi indicates a permanent blood circulation17.
Thrombophilia and a maternal immunological mechanism whose antibodies probably could to provoque the lisis of decidual cells in our case and of this mode to originate a severe premature placental detachment of normal insert placenta or a severe placental abruption. It is possible that antiphospholipid antibodies as inmunoglobulins IgG, IgM, IgA which appear in bacterial and viral infections provoqued the death of decidual and trophoblast cells damaging the structural organization of their cell membranes18
Placental villi which gets connects to the endometrium suffers furtherly a process of degeneration and finishes in this hostile ambient with detachment of the extracellular fibrinoid matrix in the thickness of the basal plate.
It seems that in chronic endometritis the placental villi acquires a neoplastic feature own of tumours. Masses of trophoblast have been seen invading the myometrium and disrupting its architecture, undergoing degeneration, nuclear pleomorphism and simulating a sarcoma. They can to invade blood vessels and to be present within lumen being confused as choriocarcinoma. Notwithstanding, trophoblast has ability to invade without assuming character of malignant tumor and this exaggerated expression of invasion as trophoblastic pseudotumor simulates a malignant tumor19.
The literature indicates that an embryochorionic insufficiency provoked by local inflammation as endometritis and insufficient cytotrophoblast invasion with secondary changes in placental villi could give rise to an early spontaneous abortion20.
In material of curettings biopsies the immunoperoxidase localization of human placental lactogen has been used as marker for the placental origin of the giant cells in "syncytial endometritis" of pregnancy21.
An epidemiologic study supports the idea that chronic deciduitis originates in preconceptional chronic endometritis rather than in ascending infection during the gestational period22. In this study, the exploration of data set developed in placentas between 22 and 32 weeks from non anomalus singleton liveborn infants; the histopathologic lesions showed in women with recurrent preterm delivery were: amnionitis, chorionitis, deciduitis, vasculitis, vessel fibrinoid necrosis, atherosis, decidual hemosiderin, histological features of placental abruption, citotrophoblast proliferation, nucleated erythrocytes, thrombosis intervillous, fibrin deposition, villous infarction, fibrosis, hypovascularity, stromal mineralization, hemorrhagic endovasculitis, decidual eosinophilia, plasma cell in basal plate and villitis of anchoring villi22.
So, endometritis perturbs the transformation of stromal cells to decidual cells provoquing in the decidualization an inestable adaptation of the trophoblast cells in the decidua during the gestation which increases the mechanisms that produces the abortion.The trophoblast not obtains good implantation and terminates degenerating in contact with the endometrium23.
No chronic villitis was seen in our case, since very few lymphocytes were noted infiltrated in the placental villi with absence of plasma cells and histiocytes24.
Syncytiotrophoblast resist infection; however, the immature cytotrophoblast of cell columns, most prominent in this trimester, is more vulnerable to infection increasing the possibility of abortion. Many cases of chronic villitis lacking a clinical history of infection or characteristic viral cytopathic effects are attributed to a maternal alloimmune response to trophoblast antigens25.
Aside from the lack of detectable placental organisms, absence of maternal or neonatal signs, symtoms, and the absence of treatment in CE, the presence of intervillositis and neutronphils should raise the suspicion for infection and consideration of histologic special stains for the detection of villitis26.
The most common findings in early abortions, under 12 weeks, were hydatid degeneration cystic, severely edematous, with atrophic trophoblast and without evidence of trophoblastic hyperplasia27. However, in our case scanty lymphocytic villitis without hydatid degeneration could to indicate the no envelopment of chromosomal anomaly. In these cases of endometritis also has been found hypermaturity and/or villous immaturity as main histological patterns28.
This illness has been reviewed recently as the second cause of maternal death in the post partum29. Pacients suffering from chronic endometritis may have an underlyng cancer of the cervix or endometrium and to be associated with cervical hypertrophy and induction of squamous metaplasia contributing in the development of cervical neoplasia among women with persistent high risk HPV infection30.By other hand,altered uterine contractility in these pacients could leads to uterine dysperistalsis as cause that impede the normal association of the placental villi to the endometrium as seen in fig.423.
In conclusion, during the endometritis an utero infected no permit the normal implantation of the villi by damage to the trophoblast which suffers degenerative changes for aggregating to the endometrium and increasing the mechanisms that provoque the abortion.
CONFLICT OF INTEREST:
The author reports no conflict of interest.
FUNDING: No declare.
ACKNOWLEDGEMENTS
I want to thank to the clinic staff of obstetrics and gynecology of the Central Hospital of Maracay,Aragua state, by the obtaining of the material of study and to our secretary Laury Gutierrez of Ovalles by typewrite the manuscript.
REFERENCES
1.- Greenwood SM, Moran JJ. Chronic endometritis: morphologic and clinical observations. Obstet Gynecol 1981;58:176-184.
2.- Kitaya K, Matsubayashi H, Yamaguchi K, Nishiyama R, Takaya Y, Ishikawa t et al. Chronic endometritis: potencial cause of infertility and obstetric neonatal complications. Am J Reprod Immunol 2016;75:13-22.
3.- Kitaya K, Takeuchi T, Mizuta S, Matsubayashi H, Ishikawa T. Endometritis: new time, new concepts. Fertil & Steril 2018;110(3):344-350.
4.- Cicinelli E, De Ziegler D, Nicoletti R, Colafiglio G, Saliani N, Resta L et al. Chronic endometritis: correlation among hysteroscopic, histologic, and bacteriologic findings in a prospective trial with 2190 consecutive office hysteroscopies. Fertil & Steril 2008;89:677-684.
5.- Kumar P, Shah NP, Singhai A, Chauhan DS, Katoch VM, Mittal S et al. Association of tuberculous endometritis with infertility and other gynecologic complaints of women in india. J Clin Microbiol 2008;46:4068-4070.
6.- Crum CP, Egawa K, Fenoglio CM, Richart RM. Chronic endometritis: the role of immunohistochemistry in the detection of plasma cells. Am J Obstet Gynecol 1983;147:812-815.
7.- Adegboyega PA, Pei Y, McLarty J. Relationship between eosinophils and chronic endometritis. Human Pathol 2010;41:33-37.
8.- Bayer-Garner IB, Korourian S. Plasma cells in chronic endometritis are easily identified when stained with syndecan-1. Mod Pathol 2001;14:877-889.
9.- Johnston-MacAnanny EB, Hartnett J, Engmann LL, Nulsen JC, Sanders MM, Benadiva CA. Chronic endometritis is a frecuent finding in women with recurrent implantation failure after in vitro fertilization. Fertil & Steril 2010;93: 437-441.
10.- Robbins JR, Bakardjiev AI. Pathogens and the placental fortress. Curr Opin Microbiol 2012; 15(1):36-43.
11.- Resta L, Palumbo M, Rossi R, Piscitelli D, Gracia FM, Cicinelli E. Histology of micropolyps in chronic endometritis. Histopathology 2012; 60(4):670-674.
12.- Groth JV. Chronic endometritis and the plasma cell, fact versus fiction. Fertil & Steril 2018;109(5):788.
13.- Benirschke K, Kaufmann P. Pathology of the placenta.4th Edition. New York: Springer-Verlag 2000;pp591-622.
14.- Lopez-Bernal A, Hausell DJ, Soler RC, Keeling JW, Turnbull AC. Prostaglandins, chorioamnionitis and preterm labor. Br J Obstet Gynaecol 1987;94:1156-1158.
15.- Naeye RL. Functionally important disorders of the placenta, umbilical cord, and fetal membranes. Human Pathol 1987; 18:680-691.
16.- Redline RW, Hassold T, Zaragosa M. Determinants of villous trophoblastic hyperplasia in spontaneous abortions. Mod Pathol 1998;11:762-768.
17.- Castejon SOC, Scucces MMG, Rivas AAE, Molina VR. Vasculogenesis en la vellosidad placentaria humana de nueve semanas. Gac Med Caracas 2002; 110(4):504-511.
18.- Castejon SOC. The effect antiphospholipid antibodies on the structure of the placental villi. Cell& Cellular Lif Sc J 2019;4(1):1-5.
19.- Kurman RJ, Scully RE, Norris HJ. Trophoblastic pseudotumor of the uterus. An exaggerated form of" Syncytial endometritis" simulating a malignant tumor. Cancer 1976; 38:1214-1226.
20.- Milovanov AP, Ozhiganova IN. Embryochorionic insufficiency: anatomic and physiologic prerequisites, rationale, definitions and pathogenetic mechanisms. Arkh Pathol 2014;76(3):4-8.
21.- Heyderman E, Gibbons AR, Rosen SW. Immunoperoxidase localization of human placental lactogen: a marker for the placental origin of the giant cells in "syncytial endometritis" of pregnancy. J Clin Pathol 1981;34(3):303-307.
22.- Ghidini A, Salafia CM. Histological placental lesions in women with recurrent preterm delivery. Acta Obstet Gynecol Scand 2005;84:547-550.
23.- Wu D, Kimura F, Zheng L, IshidaM, Niwa Y, Hirata K et al. Chronic endometritis modifies decidualization in human endometrial stromal cells. Reprod Biol Endocrinol 2017;15(1):16.
24.- Ernst LM, Crouch J, Rinder H, Howe JG. Bacterial etiology for chorionic villitis is not supported by polymerase chain reaction for 16SrRNADNA. Ped Develop Pathol 2005; 8(6):647-653.
25.- Mckenney AH. Defense and infection of the human placenta. APMIS 2018; 126:570-588.
26.- Keeling JW, Yee KT. Fetal and Neonatal Pathology.4th edition. London: Springer-Verlag 2007. Redline RW. Chapter4. Placental inflammation. Pp 90-99.
27.- Ornoy A, Salaman AJ, Ben ZZ,Kohn G. Placental findings in spontaneous abortions and stillbirths. Teratology 1981;24:243-252.
28.- Marchetti D,Vellone V, Dhimitri O,Fulchen E. Post-partum hemorrhage and malpractice claims:What can we learn from the findings of placental examination and endometrial curettage. A retrospective analysis of surgical pathology reports. Med Sci Law 2014;54(2):99-104.
29.- Ponce MP. La Endometritis Puerperal.Revision bibliografica. Rev elect portal med.com 2020;XV(4):165.
30.- Ljubin Sternak S,Mestrovic T. Chlamydia trachomatis and genital Mycoplasms:Pathogens with an impact on human reproductive health. J Pathogens 2014(183167):3.
CORRESPONDENCE:
Olivar C Castejon S. Bsc. Msc.
Laboratory of Electron Microscopy. Center for Research and analysis assistancel teaching of the nucleus Aragua (Ciadana).
Faculty of Health Sciences. University of Carabobo.
Aragua State.
Maracay. Venezuela.
Apdo 4944
Email: olivar.ciadanauc @ gmail.com